Clinical context
Schistosomiasis
 Schistosomiasis are zoonoses due to the infection by hematophagous trematodes of the Schistosoma genus, that live in the circulatory system. Six species are pathogenic for humans: S. haematobium, S. mansoni, S. japonicum (the 3 main species), S. mekongi, S. intercalatum and S. guineensis. They are responsible of three forms of infection: intestinal, urogenital or arteriovenous.
Schistosomiasis are zoonoses due to the infection by hematophagous trematodes of the Schistosoma genus, that live in the circulatory system. Six species are pathogenic for humans: S. haematobium, S. mansoni, S. japonicum (the 3 main species), S. mekongi, S. intercalatum and S. guineensis. They are responsible of three forms of infection: intestinal, urogenital or arteriovenous.
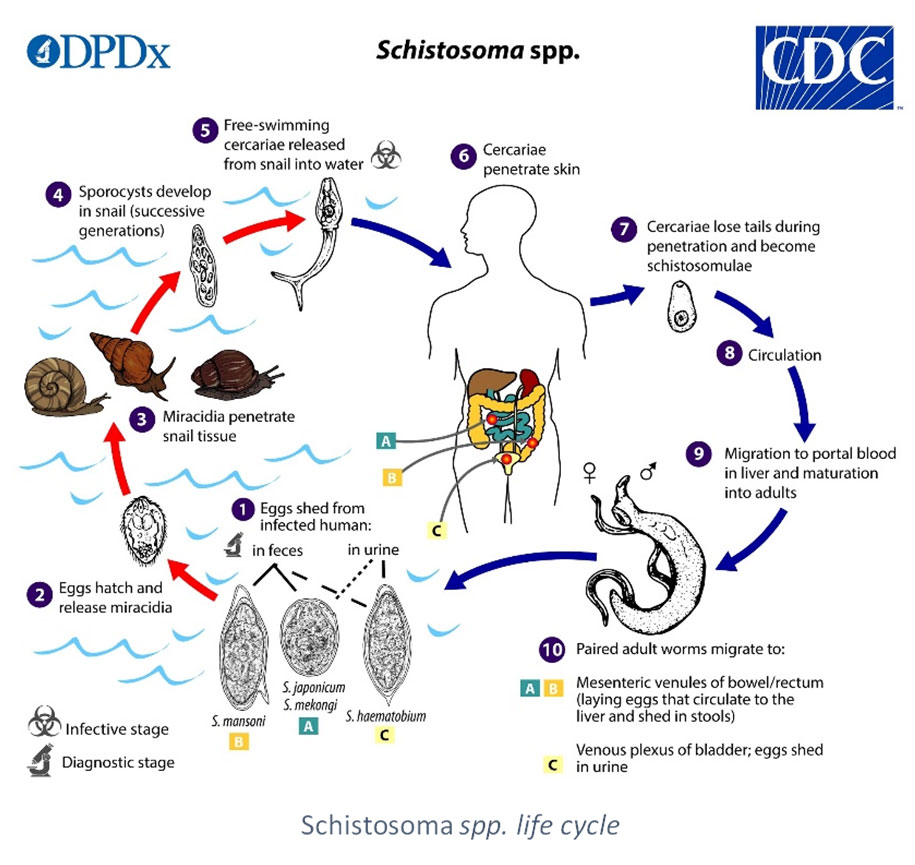
The different species have a similar life cycle. They differ by the nature of their intermediate hosts, aquatic snails which plays a determining role in the epidemiology of the disease. The different species also differ in the physiopathology of the disease in its chronic phase (urinary schistosomiasis for S. haematobium, arterioveinous for S. japonicum and S. mekongi, intestinal for the others). Every species has its own endemic region, but several species may be found in the same area.
Recent breakthroughs have shown that schistosomes can breed with worms of other Schistosoma species (Kincaid-Smith et al., 2021), leading to hybrid forms, for instance urinary schistosomiasis with S. mansoni eggs. These kinds of hybridization raise unsolved questions, including the possible reservoirs of such hybrids (Le Govic et al., 2019).
After an asexual cycle of the parasite in the snail, humans (definitive host) are infected by transcutaneous penetration of the parasite. The free-ranging infective larva (furcocercariae) enters its host during a bath in freshwater (agricultural work, ablutions…). Adult forms migrate in the veins of their hosts, where the reproduction takes place. Each specie has a specific tropism for certain veins that determines if eggs are released in feces or urine. They hatch in water to liberate the ciliated myracidiae form which infects the snail (Colley et al., 2014).
The first stage of infestation is the cutaneous penetration of the worms, which can induce a local rash called cercarian dermatitis. After several weeks occurs the invasion phase for a couple weeks (toxemia, acute schistosomiasis, Katayama fever) before the chronic phase (location varies according to the species). Clinical signs and prognosis are highly variable depending on the parasite, the individual host and the contamination conditions (massive or progressive, iterative, chronical…). It has recently been shown a higher risk of co-infection with HIV for urogenital schistosomiasis with S. haematobium (LoVerde, 2019).
The WHO estimates that 230 million people present the symptoms of the infection, and that 800 other millions are at risk. Endemic in 74 countries (Subsaharian Africa (92% of cases), oriental Mediterranea, Asia, Latin America and Caribbean), schistosomiasis are more and more often diagnosed worldwide because of migratory flows and tourism (WHO, 2020).
Since 2011, southern Europe is facing reemerging cases of schistosomiases, despite the fact that the parasite was considered eradicated in this region. First cases would be of Corsican origin, due to a new hybrid species between S. haematobium and S. bovis (which is not pathogenic for humans). These recent findings lead to several publications, and are closely followed by health organizations (Moné et al., 2015, Boissier et al., 2015, Holtfreter et al., 2014).
Diagnosis relies on epidemiology, clinical signs, direct parasitology (eggs research in urine or feces, or rectal biopsy depending on the species), imaging (ultrasonography), biological data, antigen research or serology.
Indirect immunofluorescence, indirect haemagglutination and ELISA are the most common techniques. Using natural antigens more or less purified, these techniques can lack sensitivity and specificity (Gray et al., 2011).
SCIENTIFIC REFERENCES
- Beltrame A, Guerriero M, Angheben A, Gobbi F, Requena-Mendez A, Zammarchi L, et al. Accuracy of parasitological and immunological tests for the screening of human schistosomiasis in immigrants and refugees from African countries: An approach with Latent Class Analysis. 2017 Jun; PLoS Negl Trop Dis 11(6):e0005593.
- Berry A, Fillaux J, Martin-Blondel G, Boissier J, Iriart X, Marchou B, et al. Evidence for a permanent presence of schistosomiasis in Corsica, France, 2015. 2016; Euro Surveill 21(1).
- Bevilacqua N, Pane S, Vairo F, Nicastri E, Paglia MG, Ame SM, et al. Accuracy of indirect haemagglutination and western blot assays for the detection of anti-Schistosoma antibodies in non-severe febrile patients in two Tanzanian hospitals. 2012; Scand J Infect Dis 44(6):453-8.
- Boissier J, Moné H, Mitta G, Bargues MD, Molyneux D, Mas-Coma S. Schistosomiasis Reaches Europe. 2015; Lancet Infect Dis 15 (7): 757‑58.
- Brunet J, Pfaff AW, Hansmann Y, Grogorowicz G, Pesson B, Abou-Bacar A et al. An Unusual Case of Hematuria in a French Family Returning from Corsica. 2015; Int J Infect Dis 31: 59‑60.
- Cavalcanti MG, Silva LF, Peralta RHS, Barreto MGM, Peralta JM. Schistosomiasis in Areas of Low Endemicity: A New Era in Diagnosis. 2013; Trends Parasitol 29 (2): 75‑82.
- Colley DG, Bustinduy AL, Secor WE, King CH. Human Schistosomiasis., 2014; Lancet 383 (9936): 2253‑64.
- De Laval F, Savini H, Biance-Valero E, Simon F. Human Schistosomiasis: An Emerging Threat for Europe, 2014; Lancet 384 (9948): 1094‑95.
- European Centre for Disease Prevention and Control. Rapid risk assessment: Local transmission of Schistosoma haematobium in Corsica, France –16 May 2014. ECDC; 2014
- Holtfreter, MC, Moné H, Müller-Stöver I, Mouahid G, Richter J. Schistosoma Haematobium Infections Acquired in Corsica, France, August 2013. 2014; Euro Surveill 19 (22).
- Le Govic Y, Kincaid-Smith J, Allienne J-F, Rey O, de Gentile L, Boissier J. Schistosoma haematobium–Schistosoma mansoni Hybrid Parasite in Migrant Boy, France, 2017. 2019 Feb; Emerg Infect Dis, Vol 25, Number 2.
- Moné H, Holtfreter MC, Allienne J-F, Mintsa-Nguéma R, Ibikounlé M, Boissier J, et al. Introgressive hybridizations of Schistosoma haematobium by Schistosoma bovis at the origin of the first case report of schistosomiasis in Corsica (France, Europe). 2015; Parasitol Res 114(11):4127-33.
- Noormahomed EV, Nhacupe N, Mascaró-Lazcano C, Mauaie MN, Buene T, et al. A Cross-Sectional Serological Study of Cysticercosis, Schistosomiasis, Toxocariasis and Echinococcosis in HIV-1 Infected People in Beira, Mozambique. 2014; PLoS Negl Trop Dis 8(9): e3121.
- Sulahian A, Garin YJF, Izri A, Verret C, Delaunay P, van Gool T, et al. Development and evaluation of a Western blot kit for diagnosis of schistosomiasis. 2005; Clin Diagn Lab Immunol 12(4):548–551.
- Vellere I, Mangano VD, Cagno MC, Gobbi F, Ragusa A, Bartoloni A et al. Imported human Schistosoma japonicum: A report on two cases in Filipino migrants present in Italy and a systematic review of literature. 2019; Travel Med Inf Dis, 101496.
- Wang W, Wang L, Liang YS. Susceptibility or Resistance of Praziquantel in Human Schistosomiasis: A Review. 2012; Parasitol Res 111(5):1871‑77.
THE SCHISTOSOMA ICT IgG-IgM TEST
Ease of use, fast result obtention and reliability characterize the SCHISTOSOMA ICT IgG-IgM screening test. Particularly adapted to first line laboratories and small series, this Point-Of-Care test can advantageously replace the other screening techniques.
Its excellent performance allow the isolated use of the ICT for the screening of patients, before therapeutic campaign instauration for instance, or when confirmation by Western Blot is not possible (Beltrame et al., 2017).
THE SCHISTO II Western Blot IgG TEST
The Haute Autorité de Santé (HAS) in France as well as the American Center for Disease Control (CDC) recommend the immunoblot for the confirmation of serodiagnoses (Argumentaire HAS, 2017, DPDx Schistosomiasis, 2019).
Thanks to its high performances, including in the case of infections by hybrids, Western Blot can be considered as the gold standard for the initial diagnosis of schistosomiasis (Guegan et al., 2019).
In order to meet this demand, we developed a reliable test based on the Western Blot technique. Associated with highly sensitive natural antigens (S. mansoni and S. haematobium), the SCHISTO II Western Blot IgG test is a robust confirmation technique of classical screening tests.
Our products
Our products
Order
To order a product, please send us an email to our address Order with the description of the product. We ship worldwide.
