Clinical context
Leishmaniasis
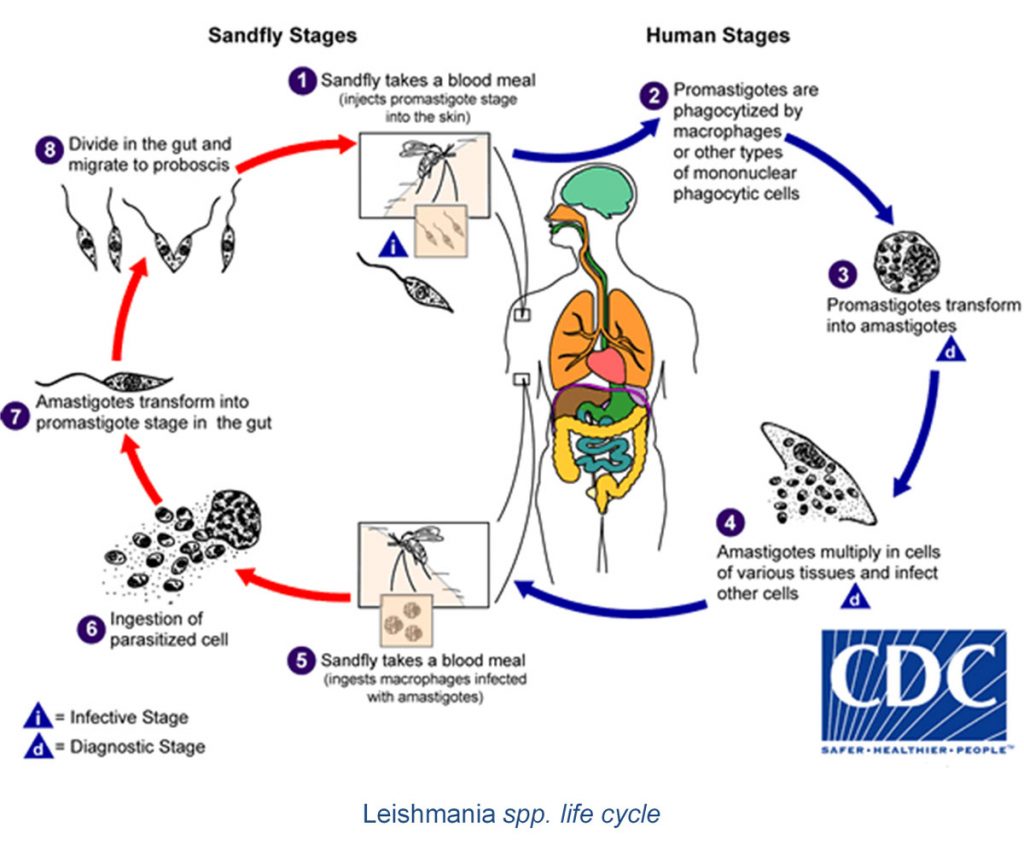 Leishmaniases are parasitic diseases of the monocyte-macrophage system, whose pathogenic agent is a flagellated protozoan of the Leishmania genus. They are transmitted at night by the female phlebotominae. The parasite, counting more than fifty different species including 20 infecting humans, has a zoonotic or anthroponotic transmission, from the vector to the mammalian reservoir (Lachaud et al., 2017).
Leishmaniases are parasitic diseases of the monocyte-macrophage system, whose pathogenic agent is a flagellated protozoan of the Leishmania genus. They are transmitted at night by the female phlebotominae. The parasite, counting more than fifty different species including 20 infecting humans, has a zoonotic or anthroponotic transmission, from the vector to the mammalian reservoir (Lachaud et al., 2017).
The parasite has two morphological stages in its life cycle: a flagellated promastigote form, extracellular and mobile, found in the digestive tube of the phlebotominae. The amastigote form, immobile and intracellular, actively multiplies within the mammalian host macrophages until their lysis, releasing them to infect other macrophages (Pace, 2014).
The disease is endemic in four continents (Africa, Latin and South Americas, Europe), with 10 to 20 million people suspected of being infected worldwide, 400,000 new cases of visceral leishmaniasis every year, and 350,000,000 people at risk of infection (Okwor & Uzonna, 2016).
Three forms of leishmaniases are described: visceral, cutaneous or mucocutaneous, asymptomatic carriers also exist. Clinical signs differ according to the species of Leishmania and the immune response of the host.
Asymptomatic forms are very common in endemic areas, where prevalence is high: 12,5% of blood donors are asymptomatic in the region of Bologna (Ortalli et al., 2019).
Cutaneous leishmaniasis, or Oriental sore, corresponds to a circular lesion at the vector sting zone. It starts as a small inflammatory papule that grows progressively.
Mucocutaneous leishmaniasis is observed in Latin America. After a facial cutaneous lesion, a granuloma grows, destroying the nasal then buccal walls and the larynx (espundia).
Visceral leishmaniasis (VL) is a slow evolution of a visceral infection, caracterised by the triad anemia, splenomegalia and irregular fever. In Europe, VL, induced by L. infantum, is endemic of several regions in France, Italie, Greece and Spain. Usually observed among children under 8 years old and in limited areas, adult (mostly aged patients) and imported cases are increasing. It is an opportunistic disease of immunodepressed patients, lethal without treatment and commonly associated with AIDS (Burza et al., 2018).
Associated to direct detection techniques (medullary sampling), serology is considered as a major tool for the diagnosis of VL (Lévêque et al., 2020a) and classical techniques (immunofluorescence and ELISA) are widely used. Nevertheless, these techniques may lack sensitivity and specificity in particular for immunodepressed or asymptomatic patients, as their immune reaction is low (Elmahallawy et al., 2014, Griensven et al., 2014, Lévêque et al., 2020b).
SCIENTIFIC REFERENCES
- Aoun O, Mary C, Roqueplo C, Marié JL, Terrier O, Levieuge A, et al. Canine leishmaniasis in south-east of France: screening of Leishmania infantum antibodies (western blotting, ELISA) and parasitaemia levels by PCR quantification. Vet Parasitol. 2009 Dec;166(1–2):27–31.
- Biglino A, Bolla C, Concialdi E, Trisciuoglio A, Romano A, Ferroglio E. Asymptomatic Leishmania infantum Infection in an Area of Northwestern Italy (Piedmont Region) Where Such Infections Are Traditionally Nonendemic. J Clin Microbiol. 2010 Jan 1;48(1):131–6.
- Cota GF, de Sousa MR, Demarqui FN, Rabello A. The Diagnostic Accuracy of Serologic and Molecular Methods for Detecting Visceral Leishmaniasis in HIV Infected Patients: Meta-Analysis. Boelaert M, editor. PLoS Negl Trop Dis. 2012 May 29;6(5):e1665.
- Deniau M, Cañavate C, Faraut-Gambarelli F, Marty P. The biological diagnosis of leishmaniasis in HIV-infected patients. Ann Trop Med Parasitol. 2003 Oct;97 Suppl 1:115–33.
- Faucher B, Pomares C, Fourcade S, Benyamine A, Marty P, Pratlong L et al. Mucosal Leishmania infantum leishmaniasis: specific pattern in a multicentre survey and historical cases. J Infect 2011 63(1):76-82
- Ferroglio E, Zanet S, Mignone W, Poggi M, Trisciuoglio A, Bianciardi P. Evaluation of a Rapid Device for Serological Diagnosis of Leishmania infantum Infection in Dogs as an Alternative to Immunofluorescence Assay and Western Blotting. Clin Vaccine Immunol. 2013 May;20(5):657–9.
- Kallel K, Ammari L, Kaouech E, Belhadj S, Anane S, Kilani B, et al. [Asymptomatic bearing of Leishmania infantum among Tunisian HIV infected patients]. Pathol Biol. 2007 Dec;55(10):521–4.
- Lachaud L, Dedet JP, Marty P, Faraut F, Buffet P, Gangneux JP, Ravel C, Bastien P, Working Group for the Notification of Human Leishmanioses in France. Surveillance of leishmaniases in France, 1999 to 2012. Euro Surveill 2013 18(29):20534
- Marty P, Lelievre A, Quaranta JF, Rahal A, Gari-Toussaint M, Le YF. Use of the leishmania skin test and western blot analysis for epidemiological studies in visceral leishmaniasis areas: experience in a highly endemic focus in Alpes-Maritimes (France). Trans R Soc Trop Med Hyg. 1994;88(6):658–9.
- Marty P, Lelièvre A, Quaranta J-F, Suffia I, Eulalio M, Gari-Toussaint M, et al. Detection by Western blot of four antigens characterizing acute clinical leishmaniasis due to Leishmania infantum. Trans R Soc Trop Med Hyg. 1995 Nov 1;89(6):690–1.
- Mary C, Lamouroux D, Dunan S, Quilici M. Western blot analysis of antibodies to Leishmania infantum antigens: potential of the 14-kD and 16-kD antigens for diagnosis and epidemiologic purposes. Am J Trop Med Hyg. 1992 Dec;47(6):764–71.
- Pomares C, Despierres L, del Giudice P, Delaunay P, Michel G, Ferrua B, et al. Western blot analysis as an aid for the diagnosis of cutaneous leishmaniasis due to Leishmania major. Trans R Soc Trop Med Hyg. 2012 Jul;106(7):452–4.
- Ready PD. Epidemiology of visceral leishmaniasis. Clin Epidemiol. 2014;6:147–54.
- Saghrouni F, Khammari I, Kaabia N, Bouguila J, Ben Abdeljelil J, Fathallah A, et al. Asymptomatic carriage of Leishmania in family members of patients with visceral leishmaniasis in Central Tunisia. Pathol Biol. 2012 Oct;60(5):e55-58.
- Solano-Gallego L, Miró G, Koutinas A, Cardoso L, Pennisi MG, Ferrer L, et al. LeishVet guidelines for the practical management of canine leishmaniosis. Parasit Vectors. 2011 May 20;4:86.
- van Griensven J, Carrillo E, López-Vélez R, Lynen L, Moreno J. Leishmaniasis in immunosuppressed individuals. Clin Microbiol Infect 2014 20(4):286-99
LEISHMANIA Western Blot IgG TEST
The French Haute Autorité de Santé (HAS) recommends the immunoblot for the diagnosis of VL and cutaneous/mucosal leishmaniases as a confirmation of the screening techniques (Argumentaire HAS, 2017).
For patients coinfected with HIV, even though DNA detection by amplification techniques are more sensitive, detection of low titers of antibodies is highly helpful for the diagnosis (Cota et al., 2012).
Thanks to its performances, the Western Blot technique can be useful as an epidemiological marker of the disease and allows a precocious diagnosis, especially with poorly evocative symptoms (Biglino et al., 2010, Pomares et al., 2012, Saghrouni et al., 2012).
In order to meet this demand, we developed a reliable test based on the Western blot technique. Associated with highly sensitive natural antigens, the LEISHMANIA Western Blot IgG test is a robust confirmation technique of classical screening tests.
The LEISHMANIA Western Blot IgG test allows the differentiation of the 14 and 16 kDa antigens, which are specific of Leishmania (Mary et al., 1992). Specificity and remarkably high sensitivity of the immunoblot technique can greatly improve the screening of visceral leishmaniasis.
Our products
Order
To order a product, please send us an email to our address Order with the description of the product. We ship worldwide.
